Article by Jason Lo Hog Tian
Graphic design by Colleen Paris
After over a year living in a different world under the COVID-19 pandemic, people are eagerly awaiting a time when they can return to normal. For most, their hopes lie in the development of a vaccine. With some vaccines in stage 3 clinical trials, many are hopeful for regulatory approval by early 2021 so we can resume our social gatherings and beach vacations. However, an approved vaccine does not mean the pandemic will be over – it is only the first hurdle. Challenges with safety, production, and distribution could mean a significant amount of time between the first vaccine approval and its widespread availability for the average person.
“An approved vaccine does not mean the pandemic will be over – it is only the first hurdle”
The purpose of a vaccine is to activate an immune response to a specific virus, resulting in long-term immunity without causing disease. When a virus enters the body, it is recognized and triggers an immune response which kills the virus as well as produces antibodies that blocks it from infecting cells and marks it for destruction.1 After being infected, the body can “remember” the virus, preventing it from causing disease if the body is re-infected and providing long-term immunity. The road to creating a vaccine is long and contains many stages, with most failing to reach final approval. Preclinical testing is the first step where vaccines are tested on animal models to see if they produce an immune response. In Phase 1, vaccines are given to a small number of people to test dosage and safety as well as to confirm if it produces an immune response. Phase 2 includes up to hundreds of people from different populations to verify safety and efficacy. Phase 3 is the last step leading to regulatory approval and involves administering the vaccine to thousands of people and examining how many get infected compared to a control group as well as identifying any rare side effects. Following the completion of Phase 3 trials, the data will be evaluated by regulating bodies and an approval decision will be made.
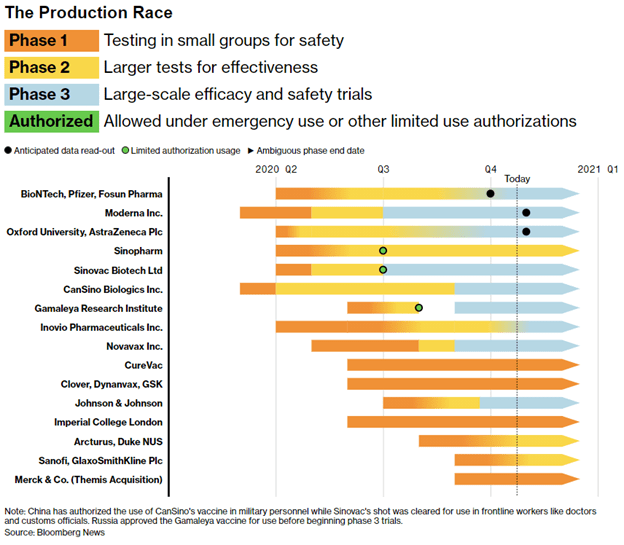
There are many companies competing to develop the first COVID-19 vaccine with a variety of methods available to activate the immune response. Figure 1 shows the major manufacturers and their progress in the vaccine production race.2 Controversially, the vaccines developed by Gamaleya Research Institute, Sinopharm, CanSino Biologics, and Sinovac have been given emergency approval for use in Russia, United Arab Emirates, the Chinese military, and for Chinese medical workers respectively, despite not completing Phase 3 clinical trials.3 This highlights the highly political nature of COVID-19 vaccine development which may prioritize being first over being effective. Nations are in a Cold War-like arms race – the Russian vaccine is even named “Sputnik V” in a nod to the first satellite launched during the space race of the 1950’s.4 However, many scientists have condemned such executive oversight, fearing a lack of safety and efficacy testing. Relying on Phase 1 or 2 data and skipping large-scale clinical trials means there is no concrete evidence that the vaccine is more effective than a placebo and does not give the opportunity to detect rare side effects that may not appear in a small sample. Fast-tracking approval seems like an obvious step to eradicating the disease quicker, however the real winner of the “vaccine race” may not be the one who completes development first, but the one who figures out the daunting challenge of distributing it to an entire population.
If 60-70% of the world have to be immune to achieve herd immunity (when a majority of people are immune so the virus becomes difficult to spread), up to 5.6 billion people would have to be vaccinated to end the pandemic.5 At a time when infrastructure is reduced due to the pandemic itself, distributing a vaccine is going to require the largest scale up in capacity we have ever seen. This problem is made even more challenging with the conditions under which a vaccine must be kept as well as the time-sensitive nature of the product.
The vaccines from Moderna Inc. and BioNTech/Pfizer/Fosun have arguably the most potential since they are RNA-based vaccines which require no culture or fermentation and can be produced rapidly. However, to keep the RNA stable it must be stored in a deep freeze, up to -80 degrees Celsius.3 This not only poses a significant problem for transportation, but for storage in pharmacies or clinics where they are most likely to be utilized. Additionally, many of the vaccines being developed may require more than one dose, doubling the burden on distribution networks and adding the extra complication of re-distributing the product to people within a strict timeframe.3 To get ahead of these problems, countries are already preordering different types of vaccine and developing the manufacturing and distribution infrastructure regardless of whether it gets approved or not, hoping to land on an effective choice. Another often unconsidered factor is that the first vaccine approved may not be the best one. While priority populations such as the elderly and healthcare workers may receive a vaccine as soon as possible, there’s no telling the length of time between the first one and the “best” one – the one you or I are more likely to receive.
These are but a few of the problems that will pose a challenge to even the most industrialized countries, not to mention areas with a lack of infrastructure and healthcare services. Unfortunately, the complex task of vaccinating most of the world is likely to highlight and reinforce existing racial and socioeconomic inequalities, further extending the time it takes to achieve widespread immunity. While the end of the pandemic could not come soon enough, pushing a vaccine through the approval process is not the solution. We must uphold strict quality control standards and realize that the true battle will be delivering an effective vaccine to the entire world. It is in all our own interests to ensure equitable access to an effective COVID-19 vaccine, for the pandemic will be over not after the first vaccination, but after the last.
References
- Callaway E. The race for coronavirus vaccines: a graphical guide. Nature. 2020;580(7805):576.
- Flanagan C, Griffin R, Langreth R. Trial Hitches Slow Covid Vaccine, Treatment Timeline: Bloomberg; 2020. Available from: https://www.bloomberg.com/features/2020-coronavirus-drug-vaccine-status/.
- Corum J, Grady D, Wee S-L, Zimmer C. Coronavirus vaccine tracker. The New York Times. 2020;5.
- Mahase E. Covid-19: Russia approves vaccine without large scale testing or published results. BMJ. 2020;370:m3205.
- Bloom BR, Nowak GJ, Orenstein W. “When Will We Have a Vaccine?”—Understanding Questions and Answers about Covid-19 Vaccination. New England Journal of Medicine. 2020.

You must be logged in to post a comment.