Understanding the cause and effect of Helicobacter pylori infection on gastrointestinal health
Article by Vritika Batura
Graphic design by Sherry An
Host-pathogen interactions are highly dynamic processes, that can include adhesion, invasion and resolution.1 The process begins with the pathogen infecting its host and ultimately ends with the host initiating an appropriate immune response to clear the infection and build immunity for future encounters.1 But what happens when the pathogen hides from the host? What mechanisms are at play? Is the host able to resolve the infection? That’s the tale of Helicobacter pylori, a gram negative spiral shaped bacterium, that infects 50% of the world population.2
Dr. Nicola Jones garnered interest in studying H. pylori during her fellowship in Paediatric Gastroenterology at The Hospital for Sick Children (Sickkids). She joined the lab of Dr. Philip Sherman to do research as part of the fellowship training with the goal of ultimately returning to the clinic as a full-time clinician. However, the intellectually stimulating environment in the Sherman lab allowed her to look at scientific problems in a new way, where she was able to combine patient care with microbiology and molecular genetics. This motivated her to pursue a PhD in this field and was the start of her journey towards becoming a well-established clinician-scientist in Gastroenterology and the Director of Integrated Physician Scientist Training Program at the University of Toronto. Her current appointments speak to her merit as a dedicated clinician-scientist and are reflective of her success and commitment to both research and patient care.

Dr. Nicola Jones
MD, PhD | Professor, Department of Pediatrics, University of Toronto | Professor, Department of Physiology, University of Toronto | Professor, Institute of Medical Science, University of Toronto | Director, Integrated Physician Scientist Training Program, Faculty of Medicine, University of Toronto | Staff Gastroenterologist, Division of Gastroenterology, Hepatology and Nutrition, Hospital for Sick Children | Senior Scientist, Cell Biology, Sickkids Research Institute
Photo Credit: Dorsa Derakhshan
During her PhD, she started exploring host-pathogen interactions. The Nobel prize winning discovery of H. pylori by J Robin Warren and Barry Marshall had gained much attention at the time. It was evident that the bacteria was highly pathogenic and had an active role in chronic gastritis.3 Dr. Jones recalls, “I really wanted to understand how the infection evades the immune system or the host responses so that you get chronic infection for life with most people not developing any complications and only ten percent developing peptic ulcer disease and less than one percent developing gastric cancer. How does this happen?”
H. pylori is a genetically diverse bacterial species and is considered the most significant risk factor for gastric cancer.2 Studies have shown that eradicating the bacteria through therapy can reduce the risk of gastric cancer.4 Current therapy to treat H. pylori infection involves the use of aggressive antibiotic treatment but antibiotic resistance is a major hurdle in H. pylori eradication.2 The real question then became, how does the bacteria acquire this resistance? Dr. Jones mentioned that during her PhD, she noticed the presence of vacuoles in gastric cells infected with H. pylori. It was later found that these vacuoles were induced by a bacterial toxin called the vacuolating cytotoxin (VacA), and these vacuoles formed a niche that permitted survival of H. pylori inside of the cells.5 Interestingly, with the use of an animal model her lab showed that mice infected with strains of H. pylori that secreted the toxin had bacteria detected within gastric cells but mice infected with strains that did not secrete vacA, did not have intracellular bacteria.6 This finding of intracellular bacteria during infection with VacA+ but not VacA- H. pylori was also seen in gastric biopsies obtained from patients. Subsequent studies showed that antibiotic treatment of mice infected with VacA- H. pylori resulted in complete eradication of the infection, whereas intracellular bacteria persisted in mice infected with VacA+ H. pylori .6 Dr. Jones explains, “Having this intracellular niche actually protects the bacteria from eradication therapy and potentially host responses. It’s a place for the bacteria to hide from clearance.”
How does this bacterial strain form these vacuoles and hide inside the cells? Dr. Jones research delineated that it inhibits a specific calcium channel, TRPML1, that is involved in endosomal trafficking.7 Her recent paper highlighted the mechanism that is at play by H. pylori (vacA+) strain. VacA inhibits the channel and disrupts endolysosomal trafficking resulting in the formation of these large vacuoles (dysfunctional lysosomes) that ultimately provide safe haven for the bacteria to hide from host immune responses.6 She is now looking into a potential drug that would activate this channel. Treatment with a small molecule TRPML1 agonist (ML-SA1), causes resolution of the large vacuoles in VacA+ infected gastric epithelial cells and human gastric organoids, resulting in formation of normal functional lysosomes and ultimately bacterial killing.6 Targeting TRPML1 could be a novel treatment to get rid of the niche that allows the bacteria to escape antibiotics and the host immune response. This could be a potential treatment to be used as part of eradication therapy and would hopefully decrease antibiotic resistance.
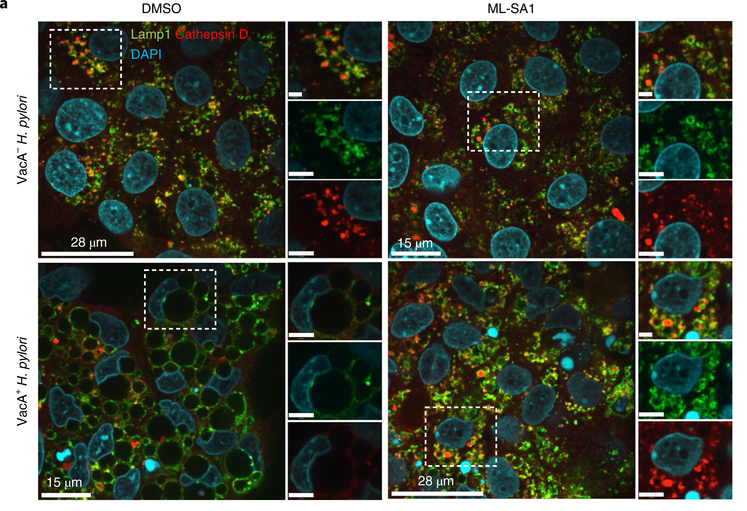
When asked how H. pylori infection leads to increased risk of gastric cancer, she explained that H. pylori forms these vacuoles only in certain cell types, specifically parietal cells. Parietal cells are the acid secreting cells of the stomach and are long-lived gastric cells. “Smart for the bacteria to hide in these cells where they can live happily for a long time and every so often get out in the lumen and infect other cells.” She mentions that if during H. pylori infection, gastric acid secretion is inhibited, it can increase the risk of gastric cancer development. Other risk factors include chronic inflammation, gastric atrophy (loss of parietal cells) and intestinal metaplasia. She hopes that studying the effects of the intracellular bacteria can shed light on mechanisms involved in gastric cancer development.
But is H. pylori all bad? She explained that studies have shown an inverse relationship between H. pylori infection and inflammatory bowel disease (IBD) and asthma. In fact, some animal studies have shown that exposure to H. pylori in young animals can provide protection against IBD and asthma. Hence, there are conflicting schools of thoughts: one believes that H. pylori must be eradicated as its pathogenic and the other believes that it might have some health benefits.
Dr. Jones believes that being a clinician-scientist provides unique opportunities to contribute to her field of gastroenterology by beginning to understand the pathophysiology of H. pylori infection. She says, “As a clinician-scientist you have a unique lens; you can identify what the clinical problems are, which allows you to ask relevant questions and then hopefully discover pathways or novel treatment options that ultimately have a positive impact on patients.” It was the opportunity, support and mentorship provided to her during her fellowship and PhD, which allowed her to pursue this direction. She considers this a privilege and has dedicated part of her career to the development of new clinician-scientists as an advocate and mentor.
References
- Jo, E-K. Interplay between host and pathogen: immune defense and beyond. Exp. Mol. Med. 2019;51(12), 1–3.
- Miller, AK, Williams, SM. Helicobacter pylori infection causes both protective and deleterious effects in human health and disease. Genes & Immunity. 2021;22(4):218-26
- Robin Warren, J., Marshall, B. Unidentifed curved bacilli on gastric epithelium in active chronic gastritis. The Lancet. 1983;321(8336), 1273–1275.
- Mégraud, F, Bessède, E,Varon, C. Helicobacter pylori infection and gastric carcinoma. Clin. Microbiol. Infect. 2015;21(11), 984–990.
- Cover TL, Blanke SR. Helicobacter pylori VacA, a paradigm for toxin multifunctionality. Nat. Rev. Microbiol. 2005;3(4), 320–332.
- Capurro, MI, Greenfield LK, Prashar A, et al. VacA generates a protective intracellular reservoir for Helicobacter pylori that is eliminated by activation of the lysosomal calcium channel TRPML1. Nat Microbiol. 2019;4(8), 1411–1423

You must be logged in to post a comment.