Article by Nayaab Punjani
Graphic design by Michie (Xingyu) Wu
Parkinson’s Disease (PD) is a neurodegenerative disorder that affects more than 100,000 Canadians.1 This condition is often characterized through motor symptoms such as tremors and non-motor symptoms including constipation and sleep disturbances.2 PD pathology involves the build-up of a protein (alpha synuclein or α-Syn) in the brain, along with the loss of dopaminergic neurons.3 Trying to find a way to reduce the expression of α-Syn, and stopping early PD progression is key to improving the patients’ quality of life. Dr. Anurag Tandon, Scientist at the Tanz Centre for Research in Neurodegenerative Diseases and Associate Professor in the Department of Medicine, Division of Neurology, uses unique approaches to address this pervasive disease.
As a cell biologist, Dr. Tandon began to understand PD through studying the role of α-Syn in vesicular trafficking and neurotransmission. Understanding of PD pathology has evolved and we now recognize the prion-like spread of α-Syn pathology between neurons. Novel therapies for PD aim to reduce α-Syn expression through either targeting its gene expression by studying viral gene knockdown vectors,4 or to decrease the concentration of the protein to reduce aggregation.
When developing immune therapies for PD, Dr. Tandon discusses the challenges of targeted delivery to the brain. Antibodies are often composed of long polypeptide chains, thus when delivered intravenously, only 0.5% pass the blood-brain-barrier (BBB) and enter the cerebrospinal fluid. However, these large proteins are very specific to the target protein. Therefore, being able to take the gene sequence for these antibodies, bridging immunotherapy with gene therapy, and express them in the brain, is a promising therapeutic avenue.
Dr. Tandon employs the use of small molecules called nanobodies. These molecules were developed through research conducted by his collaborators, David Butler and Anne Messer.5 The process of creating nanobodies involves isolating the variable domain, which recognizes α-Syn, on the heavy chain of a single chain polypeptide antibody from a human blood cell library. Human cells normally do not express single chain antibodies, however, through isolating this sequence and placing it in an expression vector, the nanobody can now bind to α-Syn, allowing it to be degraded and reduce the accumulation of this protein. In order to further improve delivery of the nanobodies using viral vectors across the BBB in PD relevant brain regions, Dr. Tandon is also exploring the use of focused ultrasound, a technique which helps large molecules to cross the BBB and can be used multiple times, to reduce α-Syn levels.
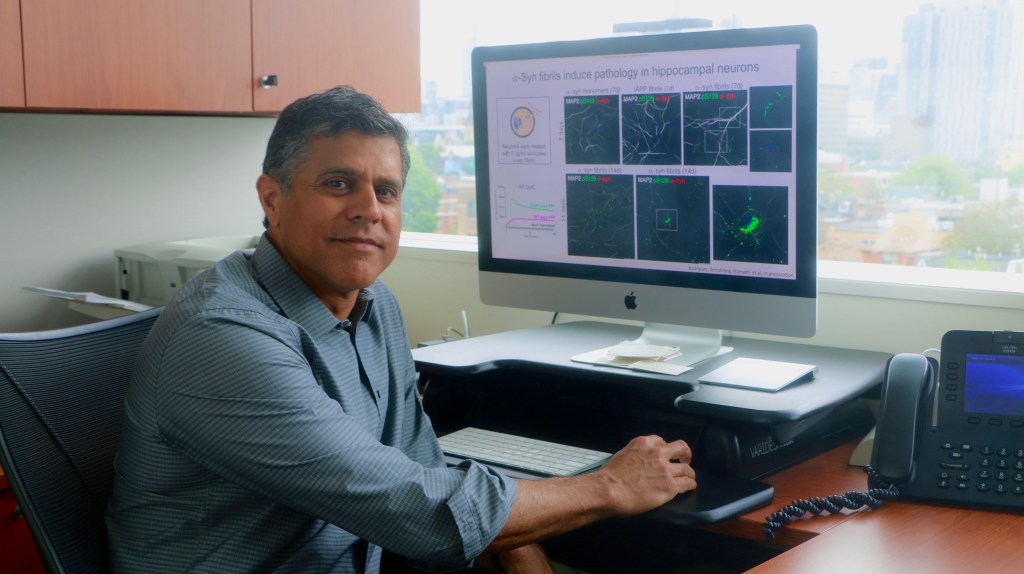
Dr. Anurag Tandon
Scientist, Tanz Centre for Research in Neurodegenerative Diseases, University of Toronto
Associate Professor, Department of Medicine, Division of Neurology
Full member, Institute of Medical Science
Photo provided by Dr. Anurag Tandon
The in vitro cell work, done by graduate student Sabrina Armstrong, demonstrates the benefit of this therapy to help reduce intracellular pathology induced by the presence of α-Syn fibrils, so his current research has shifted to examine this in animal models. Dr. Tandon explains, “The other incredible aspect of this is once there is proof of principle that it works in one model, any neurodegenerative disease could be targeted, and I do not think this is a single treatment approach, it may work in combination with other treatments as well but having a brain cell produce the therapeutic factor continuously has a certain appeal to it.” This research also opens many pathways to consider targeting α-Syn misfolding and pathological spread between cells.
While developing therapeutics for PD, it is important to use the appropriate animal models which can be challenging. “We have models that replicate aspects of the disease but we do not have an all-encompassing model”. Furthermore, “We do not fully appreciate where the disease begins in humans. There is quite a bit of research suggesting that it begins in the peripheral nervous system particularly in the gastrointestinal (GI) tract. So enteric neurons certainly show pathological changes very early on, but that may not be the case for all patients. Some patients may have a disease that starts in the brain, others may be peripheral”.
Patients generally present with PD when it has progressed to neuron loss in the brain, while the early peripheral symptoms are often missed. Thus, it is important to target this primary origin of the disease to stop progression and preserve function. Dr. Tandon is also investigating this approach by adapting animal models of enteric pathology developed by other labs, which include overexpression of α-Syn fibrils in the GI tract. Dr. Tandon aims to explore the pathology transfer from these enteric neurons to higher cortical regions of the brain, which results in dementia-like pathology of late-stage PD. Optimization of this research is ongoing and various factors will need to be considered to enhance delivery of the nanobody gene therapy vectors to peripheral neurons.
Through targeting the early underlying PD pathology in the peripheral nervous system, Dr. Tandon mentions, “Now you might have the ability to first block the spread of the pathology and then the second approach would be to see if we can rescue the existing neurons that are on the verge. We may or may not be able to rescue cells that have died completely, the distances in the brain are very challenging because to have a neuron project from the substantia nigra to the striatum, these are many centimeters, which is not a minor thing in either an animal model or humans where all the neurons are already beautifully laid out, so re-establishing connections may be a challenge, but we might be able to strengthen the existing ones”.
If your family has been impacted by Parkinson’s Disease, Dr. Tandon highlights many avenues where you can seek more information. One, is by reading magazine articles that provide updates on the current state of science in the field. Second, the Parkinson Society of Canada is a great resource to support you and assist in understanding the condition, while also connecting you with researchers. Finally, he indicates the importance of funding agencies, both government-based (Canadian Institutes of Health Research or CIHR) and private philanthropy operations like the Weston Brain Institute, which support cutting-edge research.
Dr. Tandon encourages graduate students to read about the research areas they are interested in and also examine cross-disciplinary applications. He highlights how essential collaboration is to advance scientific discoveries. Despite not having a strong virology or immunology background, through collaboration he is able to incorporate these fields into his work. He recalls, “I do not need to know how to make these things, I just need to know how I could reach the person who made them and ask them to collaborate with us on a project.” Dr. Tandon stresses the importance of humility and being open to sharing your knowledge and collaborating with scientists in other fields.
There is a lot that is yet to be explored in this field, particularly if we consider personalized medicine and assessing individual progression of PD pathology. Cross-disciplinary collaboration will be key to advancing therapies for PD and other neurodegenerative disorders.
References:
- Parkinson’s Disease [Internet]. Parkinson Canada. [cited 2022 Jun 2]. Available from: https://www.parkinson.ca/about-parkinsons/
- Symptoms of Parkinson’s [Internet]. Parkinson Canada. [cited 2022 Jun 2]. Available from: https://www.parkinson.ca/about-parkinsons/symptoms/
- Gómez-Benito, M., Granado, N., García-Sanz, P., et al. Modeling Parkinson’s Disease With the Alpha-Synuclein Protein. Front Pharmacol. 2020 Apr; 11: 356.
- Menon S, Kofoed RH, Nabbouh F, Xhima K, Al-Fahoum Y, Langman T, et al. Viral alpha-synuclein knockdown prevents spreading synucleinopathy. Brain Communications. 2021 Oct 1;3(4):fcab247.
- Messer A, Butler DC. Optimizing intracellular antibodies (intrabodies/nanobodies) to treat neurodegenerative disorders. Neurobiol Dis. 2020 Feb;134:104619.

You must be logged in to post a comment.